Class 4 Medicines Defect Information: Chelonia Healthcare Limited, Propantheline Tablets 15mg (Genesis Pharmaceuticals livery), EL(24)A/27
Chelonia Healthcare Limited has informed the MHRA that an error relating to the product description was identified in the Patient Information Leaflet (PIL) for the batches listed in this notification.
DMRC Reference Number
DMRC-30829551
Company name
Chelonia Healthcare Limited
Product name
Propantheline Tablets 15mg (Genesis Pharmaceuticals livery), PL 33414/0094
SNOMED Code
42639811000001102
| Batch Number | Expiry Date | Pack Size | First Distributed |
|---|---|---|---|
| 2308089 | Jul-26 | 112 tablets | 29-Jan-2024 |
| 2308090 | Jul-26 | 112 tablets | 07-Feb-2024 |
| 2402100 | Jan-27 | 112 tablets | 30-May-2024 |
| 2402106 | Feb-27 | 112 tablets | 30-May-2024 |
| 2402111 | Feb-27 | 112 tablets | 01-Jun-2024 |
| 2402112 | Feb-27 | 112 tablets | 01-Jun-2024 |
| 2402107 | Feb-27 | 112 tablets | 24-Jun-2024 |
| 2402108 | Feb-27 | 112 tablets | 24-Jun-2024 |
| 2402109 | Feb-27 | 112 tablets | 24-Jun-2024 |
| 2402110 | Feb-27 | 112 tablets | 24-Jun-2024 |
Active Pharmaceutical Ingredient: propantheline bromide
Brief description of the problem
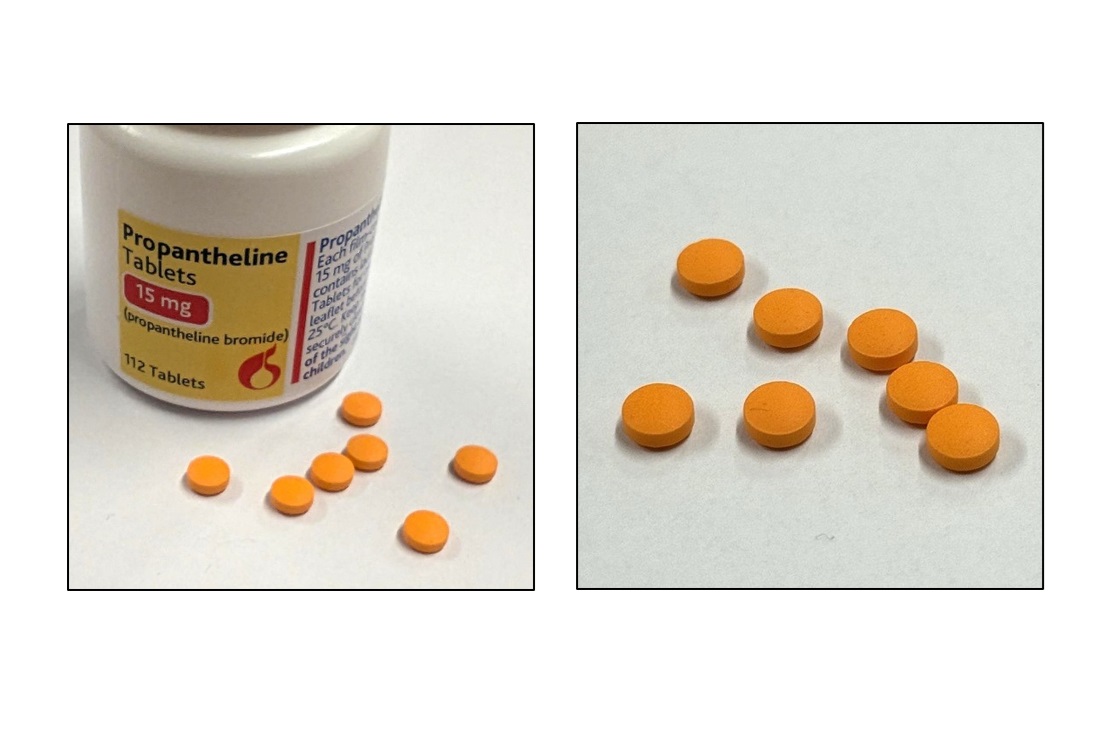
Chelonia Healthcare Limited has informed the MHRA that an error relating to the product description was identified in the Patient Information Leaflet (PIL) for the batches listed in this notification. In the PIL supplied, the product is described in Section 6 as being “pale pink in colour”, whereas the tablet is actually orange in colour.
Advice for healthcare professionals
There is no risk to product quality or safety because of this issue. Therefore the affected batches are not being recalled. Due to supply considerations, batches 2402107, 2402108, 2402109 and 2402110 have been recently distributed and were not repackaged with the updated PIL prior to distribution. Chelonia Healthcare Limited has confirmed that all future batches of the product will contain an updated PIL.
Healthcare professionals, including those involved in prescribing and dispensing, should note the correct description of the tablets as “round and convex in shape, and orange in colour”, as shown in the images below.
Advice for patients
Patients do not need to take any action. The medicine itself is not affected. Patients should continue to take medicines from these batches as prescribed by your healthcare professional.
Patients receiving the medicine via dosette boxes should continue to take the medicine in line with their prescription.
Patients who experience adverse reactions or have any questions about the medication should seek medical attention. Any suspected adverse reactions should be reported via the MHRA Yellow Card scheme.
Further Information
For stock control queries please contact info@genesis-pharma.com (Tel 020 7201 0400)
For Medical queries please contact eupvg@genreg.eu (Tel: 020 7201 0421).
Recipients of this Medicines Notification should bring it to the attention of relevant contacts by copy of this notice. NHS regional teams are asked to forward this to community pharmacists and dispensing general practitioners for information.
Yours faithfully
Defective Medicines Report Centre
10 South Colonnade
Canary Wharf
London
E14 4PU
Telephone +44 (0)20 3080 6574
Download document