MAVIS Edition 112
Published 1 November 2019
1. Product news
Isathal 10 mg/g eye drops, suspension for dogs, cats and rabbits - product defect recall alert
Known supply problems with animal medicines
Veterinary medicines will continue to be available after Brexit: Joint statement from VMD and NOAH
The VMD will launch an updated online special import application form on 7 October 2019
Top ten imported veterinary medicines - Quarterly report 1 July 2019 to 30 September 2019
Suspension of Veterinary Medicines containing the excipient Diethanolamine (DEA): Update 22 October
New national application forms to apply for, change or renew a marketing authorisation
2. Enforcement
Animal medicines seizure notice: Tudor House Animal Care Ltd
Animal medicine seizure notice: Essex Vets
Animal medicine improvement notice: Johnston Agri Supplies
Animal medicine seizure notice: Border Force, Stansted Airport
Animal medicine seizure notice: Mr Yearling
Animal medicines improvement notice: Castlehill Veterinary Clinic - August 2019
Animal medicine seizure notice: Meadowbrook Equine Clinic
3. AMR (Antimicrobial Resistance)
Sales of veterinary antibiotics halved over the past four years
3.1 Defra Antimicrobial Resistance Co-ordination (DARC) group
The DARC group met on 6 September 2019 to discuss the recent trends in antibiotic resistance (AMR) of importance to people and animals. The group received an update on antibiotic consumption data projects under development, as well as recent international collaborations. Topical presentations were given that outlined the recently published reports with contributions from the Veterinary Medicines Directorate, University of Liverpool, Royal veterinary College, Animal and Plant Health Agency and Responsible Use of Medicines in Agriculture Alliance. Summary minutes of the meeting will be published on GOV.UK in due course. Minutes from previous meetings are available.
3.2 Sales Data and Antibiotic Resistance Surveillance Report
Data for the 2018 UK Veterinary Antibiotic Resistance and Sales Surveillance (UK-VARSS) Report are currently being collated for publication later in the year. The report collates UK data on antibiotic sales from Marketing Authorisation Holders and antibiotic resistance data from the VMD’s surveillance programmes.
UK-VARSS 2018 and [previous reports-(https://www.gov.uk/government/collections/veterinary-antimicrobial-resistance-and-sales-surveillance) are available on GOV.UK.
4. Guidance updates
Export drugs and medicines: special rules
Veterinary medicine wholesale dealer’s authorisation (WDA)
Report a product defect: veterinary medicine
Authorisations to manufacture veterinary medicines
Import a medicine for veterinary use into the UK
Application form for a new national marketing authorisation
Application form to vary a national marketing authorisation
Application form to renew a national marketing authorisation
Controlled drugs: recording, using, storing and disposal
5. Surveillance
Residues of veterinary medicines in food: 2019
Veterinary Antimicrobial Resistance and Sales Surveillance 2016
Veterinary Antimicrobial Resistance and Sales Surveillance 2017
VMD Published Standards 2019 to 2020: Monitoring performance
6. Stakeholder engagement
Veterinary medicines: National authorisation application timetables from 1 April
First Joint VMD, VPC and Pharmaceutical Industry Information Event 2019
Government-funded activities to help UK businesses prepare for Brexit: veterinary medicines
6.1 Corporate news
Change to the VMD email address: Now live
Veterinary Medicines Directorate – Accessible documents policy
Veterinary Medicines Directorate – Our governance
6.2 The following staff changes took place during this quarter:
6.2 New staff
- Leah Bennett joined the VMD as a permanent AO in Licensing Administration
- Charlotte Walmsley joined the VMD as a permanent AO
- Harry Rickman joined the VMD as a permanent AO in Licensing Administration
- Amanda Gill joined the VMD as a permanent EO in Finance
- Brian Corbett joint the VMD on seconded from Dept of Education
6.3 Departing staff
- Robin Bedford
- Simon Archer
6.4 Movements within VMD
- Saul Gisborne commenced a period of TARA to EO in the IT team
- Guy Glover commenced a period of TARA to EO in the International team
- Rabia Sakhi commenced a period of TARA to EO in the EU Exit team
- Pavitra Tanwar returned to the VMD following maternity leave
- Debbie Austin commenced a period of maternity leave
- Adriana Chirilov returned to the VMD following a period of maternity leave
- Jennifer Underwood returned to the VMD following a period of maternity leave
6.3 Organogram as at 31 October
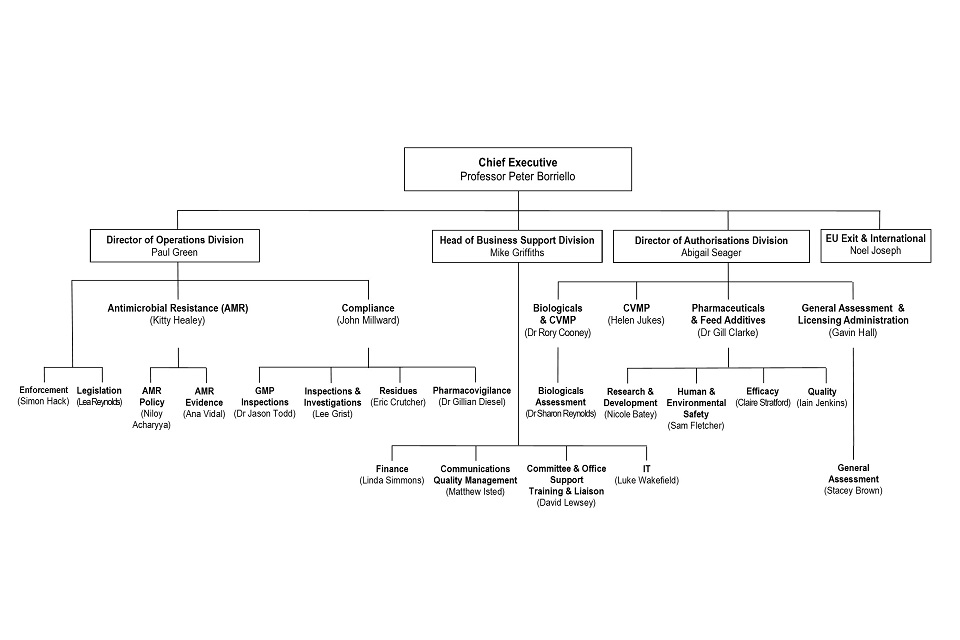
VMD Organogram as at 31 October 2019
