Medicines and Healthcare products Regulatory Agency
Regulatory Agency
- Alerts, recalls and safety information: medicines and medical devices
- Drug Safety Update
- Yellow Card: Report a problem with a medicine or medical device
- Marketing authorisations and licensing guidance
- Product information about medicines
- Regulating medical devices
- Latest information for patients
- About MHRA
- All MHRA services and information
Featured
Press release
Two arrested during the MHRA’s largest ever seizure of unlicensed weight loss medicines
The raid on a Northamptonshire estate dismantled what is believed to be a major illicit manufacturing and distribution operation

Press release
Rilzabrutinib authorised to treat adults with immune thrombocytopenia when prior treatments have been insufficient
As with any medicine, the MHRA will keep the safety and effectiveness of rilzabrutinib under close review.

Press release
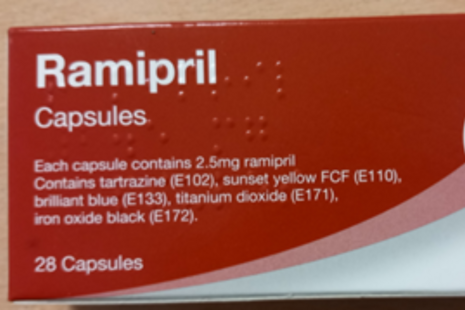
Precautionary recall of blood pressure medication (Crescent Pharma Limited Ramipril 2.5mg capsules) after packaging error
Patients taking Crescent Pharma Limited Ramipril 2.5mg capsules are advised to check their packaging for batch number GR155023 and contact their pharmacy if any blister strips inside are labelled as Ramipril 10mg.
News story
MHRA welcomes Jason Bonander as he starts his role as Chief Digital and Technology Officer
Jason is an experienced technology leader with over 25 years working in tech leadership and health informatics.

News story
Don’t let this heatwave affect your medicines: Three important tips from the MHRA
Essential advice on protecting your medicines during extreme heat and staying safe.

Press release
Landmark new plans bring treatments for rare diseases a step closer
The MHRA launches public consultation on new framework that could lead to earlier licensing for therapies for up to 3.5 million people in the UK.

Latest from the Medicines and Healthcare products Regulatory Agency
Subscriptions
What we do
The Medicines and Healthcare products Regulatory Agency regulates medicines, medical devices and blood components for transfusion in the UK.
MHRA is an executive agency, sponsored by the Department of Health and Social Care.
Follow us
Documents
Transparency and freedom of information releases
Our management










Contact MHRA
General enquiries
10 South Colonnade
London
E14 4PU
United Kingdom
Telephone
020 3080 6000
Fax
020 3118 9803
Office hours are Monday to Friday, 9am to 5pm.
Media enquiries
MHRA10 South Colonnade
London
E14 4PU
United Kingdom
Telephone (including out of hours):
020 3080 7651
Make a Freedom of Information (FOI) request
- Read about the Freedom of Information (FOI) Act and how to make a request.
- Check our previous releases to see if we’ve already answered your question.
- Make a new request by contacting us using the details below.
Freedom of Information
10 South Colonnade
London
E14 4PU
United Kingdom
