Abdominal aortic aneurysm screening: ultrasound equipment quality assurance guidance
Updated 17 April 2025
Applies to England
1. Overview
The main aim of the NHS Abdominal Aortic Aneurysm (AAA) Screening Programme is to reduce aneurysm-related mortality through the early detection, appropriate monitoring and treatment of aortic aneurysms.
The NHS AAA Screening Programme invites men for an abdominal ultrasound scan during the year (1 April – 31 March) they turn 65. The screening scans are performed by qualified screening technicians who follow a scanning protocol developed by the NHS AAA Screening Programme. This requires measurement of the aorta in transverse and longitudinal planes.
This publication provides guidance on the quality assurance of ultrasound equipment used in the NHS Abdominal Aortic Aneurysm (AAA) Screening Programme.
It is recognised that access to formal ultrasound quality assurance (QA) across England is variable, but equipment used in national screening programmes must be quality assured. Most equipment faults can be detected by visual inspection and simple tests.
This guidance sets out evidenced-based methods for the testing and monitoring of ultrasound scanners that can be undertaken by screening staff between annual assessment by medical physics departments or any other qualified provider. It also recognises that, with appropriate training and monitoring, screening technicians can perform monthly QA checks.
2. Screening equipment used in the NHS AAA Screening Programme
The 3 systems that can be bought for use in the programme are:
- Mindray – M9
- MIS – HM70 Evo
- Esaote – MyLab Omega
These systems are equipped with harmonic and compounding imaging modes that are normally switched on for screening. These should be disabled for the sensitivity and noise tests outlined in this document. Pre-existing scanner models may still be in use by a small number of services. Future equipment evaluation projects may lead to the introduction of new ultrasound models. The equipment QA policy will apply to all new systems.
3. Scope
QA of ultrasound equipment is essential to ensure safety, correct functioning of equipment, and the accuracy and reproducibility of electronic calliper diameter measurements [footnote 1] [footnote 2].
More information is provided by:
- The Institute of Physics and Engineering in Medicine policy statement on the role of the clinical scientist in medical ultrasound
- The Society of Radiographers guidelines for professional ultrasound practice
- British Medical Ultrasound Society guidelines for regular quality assurance testing of ultrasound scanners by sonographers.
Historically, regular performance of ultrasound equipment was assessed using tissue equivalent test objects. However, there are a number of limitations to this approach:
- Tissue equivalent test objects are expensive, can deteriorate with age and staff with expert knowledge are required to perform the testing.
- Modern ultrasound scanners use digital electronics to produce ultrasound images and there are no moving parts within the transducer and consequently no calibration adjustments that the operator can perform.
However, you can monitor the performance of the equipment using a range of simple routine tests. If objective testing demonstrates significant deterioration, this can also provide evidence for the replacement of older equipment.
This document is based on recommendations published by the Institute of Physics and Engineering in Medicine [footnote 1] and the British Medical Ultrasound Society [footnote 2].
The guidance comprises 4 specific elements:
- Acceptance testing on delivery of new equipment to be performed by the regional medical physics department or any other qualified provider
- Safety checks before each scanning session
- Monthly performance tests to be carried out by internal quality assurance (IQA) lead, appropriately trained clinical skills trainer (CST) or appropriately trained screening technicians
- Annual performance checks conducted by the regional medical physics department in line with local trust policy of monitoring the performance of ultrasound equipment.
Where services are unavailable, the equipment manufacturer as part of a service contract or any other qualified provider can undertake this assessment.
4. Acceptance testing
Acceptance tests must be completed before new ultrasound systems enter use into the local screening service. These should include the following checks:
-
The scanner will be checked for electrical safety by the medical physics department and sticker attached to indicate when the next check is due.
-
The equipment must be logged onto an appropriate medical equipment asset register and appropriate stickers attached to the equipment to identify this has been completed.
-
An assessment of the accuracy of electronic measurement callipers, including axial and lateral resolution, should be assessed using either an open topped test object or tissue equivalent test object. The overall penetration of the system should be assessed using an AAA specific preset with harmonic imaging and compound imaging disabled. These tests should be performed by the local medical physics department or, if this is not possible, by the equipment supplier at installation. A record of the values must be retained for future comparison by the screening programme.
-
Image uniformity should be assessed in line with IPEM report 102 and BMUS guidelines.
-
A baseline sensitivity test should be performed by imaging dry in air as recommended by IPEM Report 102 and BMUS guidelines. Measurement of the depth of reverberation lines is a proxy for sensitivity. It is recommended that harmonic imaging and compound imaging should be turned off for this test. The output and gain settings are recommended to set to maximum. The time-gain compensation controls should be set in the central position (a click is normally felt when the slider is moved into the centre). A single focus should be positioned in the upper image close to the probe.
A depth setting between 6cm and 8cm, dependent on the system, should be adequate to accommodate the reverberation pattern. This measurement is a baseline for subsequent tests. IPEM report 102 recommends that the acceptable range for future tests is ± distance to the next reverberation line.
-
A record of the pre-sets and control settings used during testing and images must be retained for future testing and reference to ensure that the same settings are used for QA of the equipment throughout its lifetime. Reports and documentation will be retained by the local screening service.
-
It is recommended that all local screening services have service contracts in place. Ideally these should provide preventative maintenance visits on an annual basis.
5. Safety checks
Before each screening session the operator will do the following checks:
-
Check that the scanner is clean and there is no contamination from gel or accumulation of dust or other debris on the surfaces or around controls.
-
Check the scanner for any visible damage such as cracks to the casing or damage to control buttons, switches and the keyboard or screen. Pay particular attention to identifying cracking around handles and corners of the display screen.
-
Check the integrity of the mains cable and if present, power transformer. Make sure there are no breaks in the cable outer insulation and, in particular, that there is no kinking or breaks in the cable at the point where it joins the mains plug, transformer or the ultrasound machine. If any damage is identified the machine must be removed from service and the fault reported and rectified.
-
Check the integrity of the transducer cable, connector and transducer head. This includes breaks in the cable or damage, cracks or splits, or irregularity in the lens surface of the transducer face (Figure 1). If there is damage to the transducer face this could result in electrical risk with gel or water ingress to the transducer elements. If such damage is identified, the scanner must be taken out of service for further evaluation.

Figure 1. Inspection of the transducer face demonstrates an obvious area of damage shown by the arrow. The probe should be removed from use and assessed by the medical physics department, equipment manufacturer or maintenance provider
-
When the scanner is switched on, look for any error messages or obvious abnormalities such as a flickering screen or excessive noise in the image that might indicate a problem.
-
Ensure the correct imaging pre-set has been selected and that the B-mode grey scale bar at the side of the image display can be fully visualised to display dark grey to black at one end and peak white at the other with a smooth graduation between the two ends.
-
Look at the uniformity of the dry in air reverberation pattern to check for irregularity that might indicate element drop out or transducer damage such as obvious disruption of the reverberation pattern (the bright parallel lines running across the image). These faults may be evident during imaging (Figure 2).
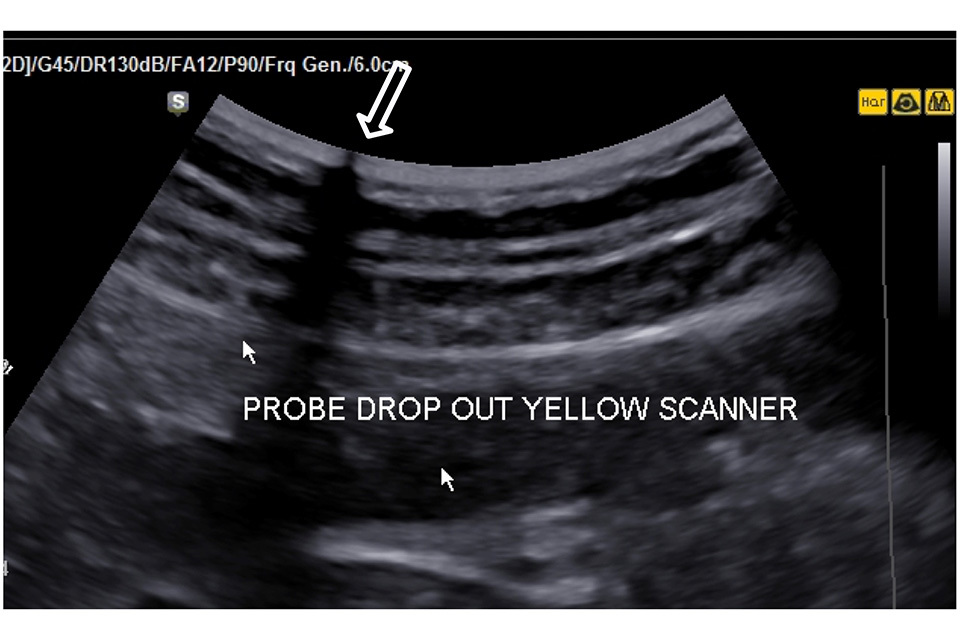
Figure 2. There is a transducer transmission fault at the point shown by the large arrow resulting in a significant loss of image along the transmission line. This probe should be removed from service and repaired or replaced
-
A log of faults, actions and outcomes should be recorded and retained by the local screening service for each scanning system.
-
At the end of each scanning session, make sure that the scanner and cables are completely clean using manufacturer recommended cleaning wipes and agents.
6. Monthly testing
Screening technicians can undertake monthly equipment QA testing but only if this has been agreed by the local screening service following training and assessment. The training and assessment must be conducted by the local screening staff who currently undertake equipment QA. This would normally be the IQA lead, CST or medical physicist, or a combination of these staff.
The criteria outlined below must be fulfilled:
-
The screening technician must not be a trainee. A screening technician must have completed and passed the Health Screener Diploma (HSD). A screening technician who qualified before the HSD can be trained to undertake monthly testing of equipment as set out in this document.
-
A formal half day training session must be provided at local level. This should include theory and practical teaching supported by reference material. An ultrasound system must be available in the training session to demonstrate the QA procedure, including machine settings and documentation of measurements.
At the end of the session, the screening technicians must pass an assessment of practical competence conducted by the IQA lead, CST or medical physicist. The screening technician must demonstrate that they can successfully undertake and document all tests listed in the monthly equipment QA schedule. This must be repeated at least 3 times during the same assessment session. This will allow the screening technician to demonstrate that they can obtain consistent results. The scanner must be switched off, then switched back on and set to a standard AAA pre-set between each testing cycle. This will allow the screening technician to demonstrate that they can correctly configure the scanner settings for the equipment QA process.
The IQA lead, CST or medical physicist is responsible for signing a document of competence, which must be retained by the local screening service.
If the screening technician is not able to demonstrate consistent measurements within the 3 cycles, further training and assessment is required.
Following satisfactory assessment and sign-off of competence, the IQA lead, CST or medical physicist must:
- observe the screening technician undertaking their first monthly equipment QA session to provide support and feedback
- review the results of subsequent monthly equipment QA tests performed by the screening technician to be aware of any equipment problems
- undertake an observational assessment of the screening technician performing equipment QA at least once every 6 months to provide feedback
- identify any subsequent sub-standard performance and take appropriate action.
Local screening services must document whether individual screening technicians have been trained to undertake equipment QA and document the observational assessment by the IQA lead, CST or medical physicist. Screening technicians can include the training in their continuing professional development portfolio.
The IQA lead, CST or appropriately trained screening technician will inspect the equipment on a monthly basis and retain a log of findings recordings, faults and actions retained.
Monthly testing includes imaging tests to be performed using the same scanner settings as initial acceptance testing. Harmonic, imaging, compound imaging and any other advanced image processing should be disabled, and the overall transducer output and gain set to maximum. The time gain compensation (TGC) sliders should be set to the mid position and a single focus point positioned at the most superficial level within the image screen close to the probe. The viewing and ambient light conditions should be the same for all monthly testing sessions.
The following tests must be undertaken to assess scanner and transducer integrity and, sensitivity and performance:
- The scanner and equipment should be checked as recommended before daily use (see checklist above).
- Image uniformity should be tested as recommended in by IPEM report 102 and BMUS guidelines. Problems such as axial banding may indicate a transmission or reception fault.
- Transmission and element faults can be checked by running a paper clip, held at 90 degrees to the long axis of the surface of the probe following application of a very thin layer of coupling gel. Look for any loss of echoes as the clip is moved across the transducer face. This can be more difficult to spot at the sides of the image sector.
- Perform a sensitivity test by imaging dry in air as recommended by IPEM report 102 and BMUS guidelines. Images or measurements should be recorded for comparison with previous tests, including the acceptance test.
- Perform an electronic noise assessment test as recommended in IPEM report 102 and BMUS guidelines.
- Check when the next electrical safety check is due. This is normally shown on tags attached to the scanner or recorded in the equipment log.
The local screening service must retain reports and documentation for each scanner.
7. Annual testing
Annual assessment of the systems is performed by the medical physics department, by the equipment manufacturer as part of preventative maintenance or by any other qualified provider using an appropriate test object in line with IPEM report 102.
Annual testing includes:
- measurement accuracy of electronic callipers including axial and lateral resolution
- penetration assessment
- assessment of transducer integrity general equipment inspection and electrical safety.
The local screening programme retains reports and documentation. Local policies may require more frequent inspections.
The performance and sensitivity of ultrasound machines can be monitored by regular testing as outlined in this document. This will also help identify deterioration that will support decision making regarding the need for replacement equipment that can be reported to programme boards.
7.1 Equipment logs
It is important that equipment checking and testing logs are kept up to date and maintained as local screening services may be called upon to provide these as evidence that appropriate checking processes have been undertaken and acted upon, as part of the ongoing quality assurance programme delivered by NHSE.
7.2 Reducing the risk of equipment damage
Careful handling and frequent inspection of ultrasound scanners will reduce the risk of damage and help to identify potential problems.
- Keep equipment clean paying particular attention to the controls and trackball for any ultrasound gel ingress as this can degrade function. Always follow the manufactures advice and local infection control policies.
- Make sure that mains cables and transducer leads are not wound too tightly, especially close to plugs, transformers, and the transducer connector as this can stress the wiring or cause the outer insulation to split.
- Inspect the probe connector before attaching it to the machine. It is possible for foreign objects such as a staple to lodge on the connector pins which cause damage to the pins as the connector is inserted into the machine.
- If the probe connector is not fitting easily to the scanner do not attempt to force a connection. Inspect the scanner and connector for damage or foreign objects.
- When opening the display screen, lift the display lid from its upper centre rather than the outer edges as this reduces stress on the screen and hinges.
- When carrying the scanner, try and pick it up by gripping the centre of the handle rather than the outer edges as this reduces the risk of stress cracks between the handle and main body of the scanner.
- Don’t place the transducer on the edge of a trolley or table, they are more likely to fall on the floor and sustain damage.
- Avoid trailing wires where they can become a trip hazard and lead to equipment being damaged.
-
IPEM Report 102 (2010) Quality Assurance of Ultrasound Imaging Systems. ISBN-10: 1903613434 ↩ ↩2
-
Dudley N, Russell S, Ward B, Hoskins P; BMUS QA working party. BMUS guidelines for the regular quality assurance testing of ultrasound scanners by sonographers. Ultrasound 2014; 22: 8-14. ↩ ↩2
